|
Thirdly, a smaller particle size also reduces the diffusion pathway of Li + ions to the interior of the particle, which leads to a greater capacity at higher charge/discharge rates and therefore to a larger power density. Secondly, nano-sized particles have larger surface areas and exhibit superior charge transfer kinetics. A larger effective contact surface with the electrolyte means a greater probability to drain Li + ions from the electrode, which increases the power density of the cell. One is the increase of the effective contact area of the powder with the electrolyte. The performance of electrode materials for Li-ion batteries reached today is the result of intensive research to reduce the size of particles to the nanoscale for three main reasons. Taking into account the molar concentration of the compounds in the Li-rich electrode, the overall theoretical capacity is expected to be 378 mAh Q th of Li 2MnO 3 and LiNi 0.5Mn 0.5O 2 are 458 mAh As an example, let us consider the theoretical capacity of the Li-rich layered material Li 1.2Ni 0.2Mn 0.6O 2 or 0.5(Li 2MnO 3) mol −1), and Z, N A, and e represent the number of electrons involved in the reaction, Avogadro’s number, and electronic charge, respectively.g −1), M w is the mass of the correlated component (g.Where Q th is the theoretical specific capacity (mAh The theoretical capacity of a given electrode material, which influences the C-rate estimations, is calculated by the following equation: One of the prospective solutions for the preparation of electrodes with high power density is the choice of nanocomposite materials because the geometric design of the insertion compound is a crucial intrinsic property from the viewpoint of structural stability and low kinetics of ions in oxide. Achieving high rate rechargeable Li-ion batteries depends ultimately on the dimension of the active particles for both negative and positive electrodes. LiCoO 2 is used as a cathode and graphite as an anode.Īs the transition metal entering the composition of the active element of the cathode is oxidized and reduced during the cell charge and discharge, respectively (Equation (1)), the cathode is primarily involved in the cathode process and then in the electrochemical performance of the cell, i.e., potential, specific capacity, energy density, rate capability, etc.Ĭonventional rechargeable Li batteries exhibit rather poor rate performance, even compared with old technologies such as lead-acid. Note that in the case of LiB with the graphite/TMO configuration, the limiting factor comes from the cathode side, in which the redox reaction is described by: Consequently, the effectiveness of a lithium-ion cell is dependent on the availability of crystallographic sites for hosting Li + ions, in other words on the insertion mechanism and thereby on the transport properties of ions and electrons in both electrode materials. During the charge of the LiB charges, the Li + ions are extracted from the cathode and inserted into the anode, while the electrons pass through the outer circuit (load). The LiB prototype was composed of graphite as a negative electrode (often named anode) and a transition-metal oxide (TMO), i.e., LiCoO 2, LiMn 2O 4, etc., as a positive electrode (often named cathode), separated by the electrolyte that provides a transport medium for ions ( Figure 1). 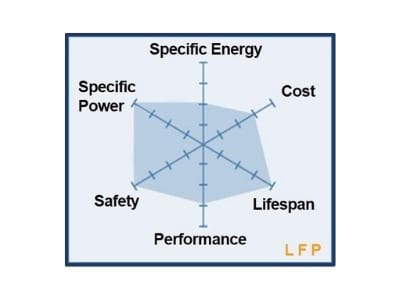
Lithium-ion batteries (LiBs) are generally composed of two electrode compounds having an open structure, which act as host frameworks for the insertion/de-insertion of Li + ions and are the place of charge transfer.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
